April 14 – Suzhou, China and Mölndal, Sweden
Suzhou Ribo Life Science (6938.HK) and its subsidiary Ribocure Pharmaceuticals AB, (together “Ribo”) today announced the successful submission of a Phase 2b Clinical Trial Application (CTA) to the European Medicine Agency (EMA) for vortosiran (RBD4059), the world’s first and most advanced siRNA therapy targeting Factor XI (FXI), for stroke prevention in atrial fibrillation (SPAF). The submission marks an important milestone in the clinical development of siRNA-based approaches to anticoagulation.
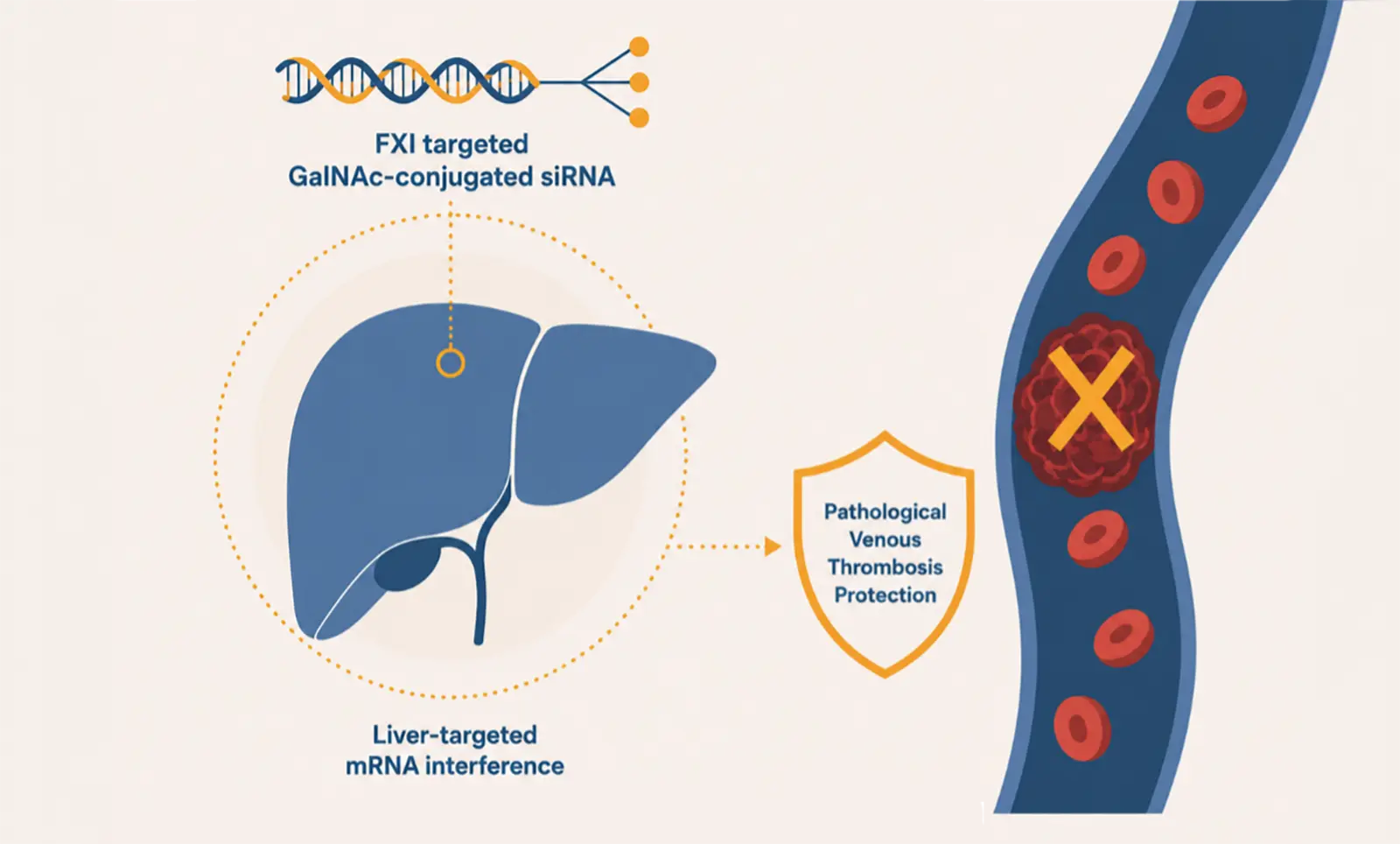
Vortosiran is designed to achieve selective and durable suppression of FXI, thereby inhibiting FXI clotting activity, with the potential to provide robust thrombotic protection with high compliance to therapy, while reducing bleeding risk and addressing a critical unmet medical need to improve benefit-risk balance compared to current anticoagulant therapies.
This study for SPAF, coded ORBIT‑XI‑AF, forms part of Ribo’s broader clinical program encompassing multiple planned Phase 2b studies across multiple thrombotic cardiovascular indications. Together with the recent completion of Ph2a study in coronary artery disease (CAD), Vortosiran has solidified its position as the most advanced siRNA‑based FXI inhibitor in global clinical development.
Stroke in AF remains a major global public health challenge, with millions of patients remaining untreated or inadequately treated due to bleeding concerns associated with current anticoagulant therapies. FXI siRNA inhibition represents a novel therapeutic approach with the potential to improve the benefit–risk profile of anticoagulation and address a key unmet need in this patient population.
“Today’s CTA submission represents a landmark achievement for Ribo and for patients who lack optimal anticoagulation options,” said Dr. Anders Gabrielsen, Chief Medical Officer. “As the pioneer in siRNA-based FXI-targeted therapeutics, we are advancing vortosiran with urgency to address the significant unmet need in SPAF. This milestone validates our proprietary siRNA platform and reinforces our leadership in next-generation anticoagulation therapies.”
Ribo (6938.HK) is committed to accelerating the global clinical development of vortosiran and building a leading franchise of RNA‑based therapies for cardiovascular and thrombotic disorders, delivering long-term value to patients, healthcare providers, and shareholders alike.