Mölndal, Sweden, Kunshan, China and Ingelheim, Germany – 3 January 2024
Suzhou Ribo Life Science Co. Ltd. and Ribocure Pharmaceuticals AB (Ribo) today announced a collaboration with Boehringer Ingelheim to develop novel treatments for nonalcoholic or metabolic dysfunction-associated steatohepatitis (NASH/MASH). The partnership brings together Ribo’s leading experience in the discovery and clinical development of small interfering RNA (siRNA) therapeutics with Boehringer’s commitment to improving the lives of people living with cardiovascular, renal and metabolic diseases (CRM).
More than 440 million people worldwide are estimated to live with NASH, an inflammatory liver disease that is caused by accumulation of fat in the liver. Over time, NASH causes scar tissue formation, which in many cases leads to liver cirrhosis and related serious complications, including liver failure or liver cancer. Currently there are no approved medicines for NASH and there is an urgent need for new effective treatment options that can prevent NASH progression and maintain and restore liver function.
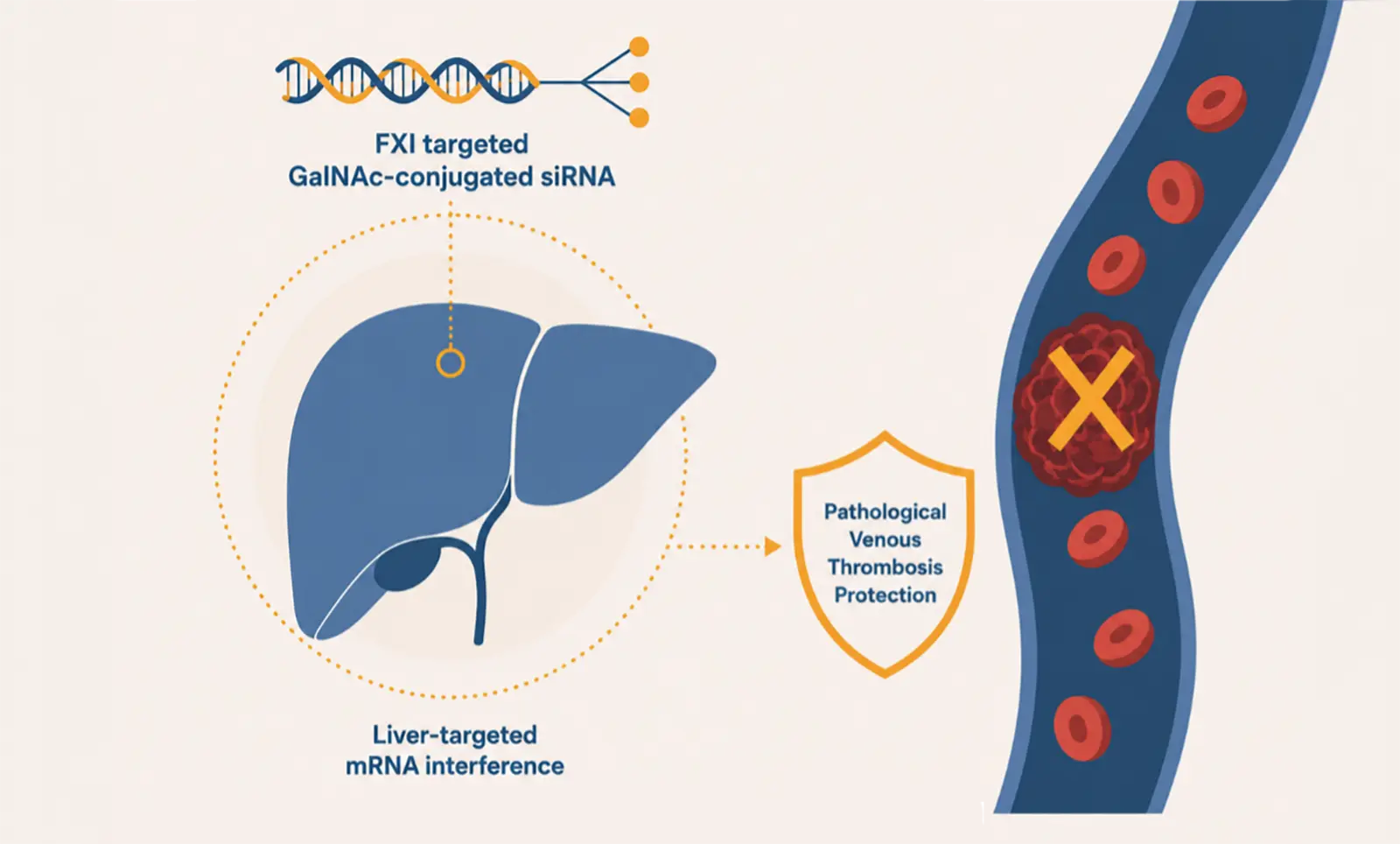
Ribo’s cutting edge RIBO-GalSTAR™ platform enables the development of RNAi therapeutics targeting disease-causing genes specifically in hepatocytes by silencing their messenger RNAs (mRNAs). This approach has the potential to treat diseases addressing previously inaccessible drug targets.
“We look forward to working with the scientists from Ribo to develop new breakthrough medicines for patients with NASH,” said Søren Tullin, Ph.D., Senior Vice President and Global Head of Cardiometabolic Diseases Research, Boehringer Ingelheim. “This new partnership is part of our commitment to collaborate with peers worldwide to address the interconnected nature of CRM diseases. Our goal is to develop the next wave of innovative medicines that will lead to a holistic health gain for patients.”
Zicai Liang, CEO of Ribo Life Science said, “This collaboration represents a strong endorsement of the many years of investment and scientific discovery built into Ribo’s platform. We are very pleased about the opportunity to work with Boehringer Ingelheim to develop new solutions for people living with NASH.”
Li-Ming Gan, CEO of Ribocure commented “We look forward to working with Boehringer Ingelheim building on their unmatched experience and understanding of CRM conditions to continue their track record of delivery of breakthrough medicines in this space.”
Under the terms of the agreement Ribo will receive an upfront payment. In this multi-target collaboration, Ribo is entitled to receiving success-based milestones for clinical, regulatory, and commercial success and tiered royalties with an overall deal value that exceeds USD 2 billion.
About Boehringer Ingelheim
Boehringer Ingelheim is working on breakthrough therapies that transform lives, today and for generations to come. As a leading research-driven biopharmaceutical company, the company creates value through innovation in areas of high unmet medical need. Founded in 1885 and family-owned ever since, Boehringer Ingelheim takes a long-term, sustainable perspective. More than 53,000 employees serve over 130 markets in the two business units Human Pharma and Animal Health. Learn more at www.boehringer-ingelheim.com
About Suzhou Ribo Life Science Co. Ltd. and Ribocure Pharmaceuticals AB
Suzhou Ribo Life Science Co. Ltd. is an innovative clinical stage R&D company devoted to the development of nucleic acid drugs and related products based on the RNA interference (RNAi) technology. With its innovative R&D capabilities with vertically integrated technological platforms, Ribo has built a strong product pipeline, aiming to make contribution to the treatment of serious diseases with unmet medical needs. As a subsidiary of Ribo Life Science Co., Ltd., Ribocure Pharmaceuticals AB is dedicated to globalized development of life-saving oligonucleotide therapies, with focus on development of assets and pipeline as well as new target ideas and on building innovative capacities to conduct clinical trials and developing siRNA drugs to address real unmet medical needs globally. For more information, please visit www.ribolia.com and www.ribocure.com