May 11 2026 – Suzhou, China and Mölndal, Sweden
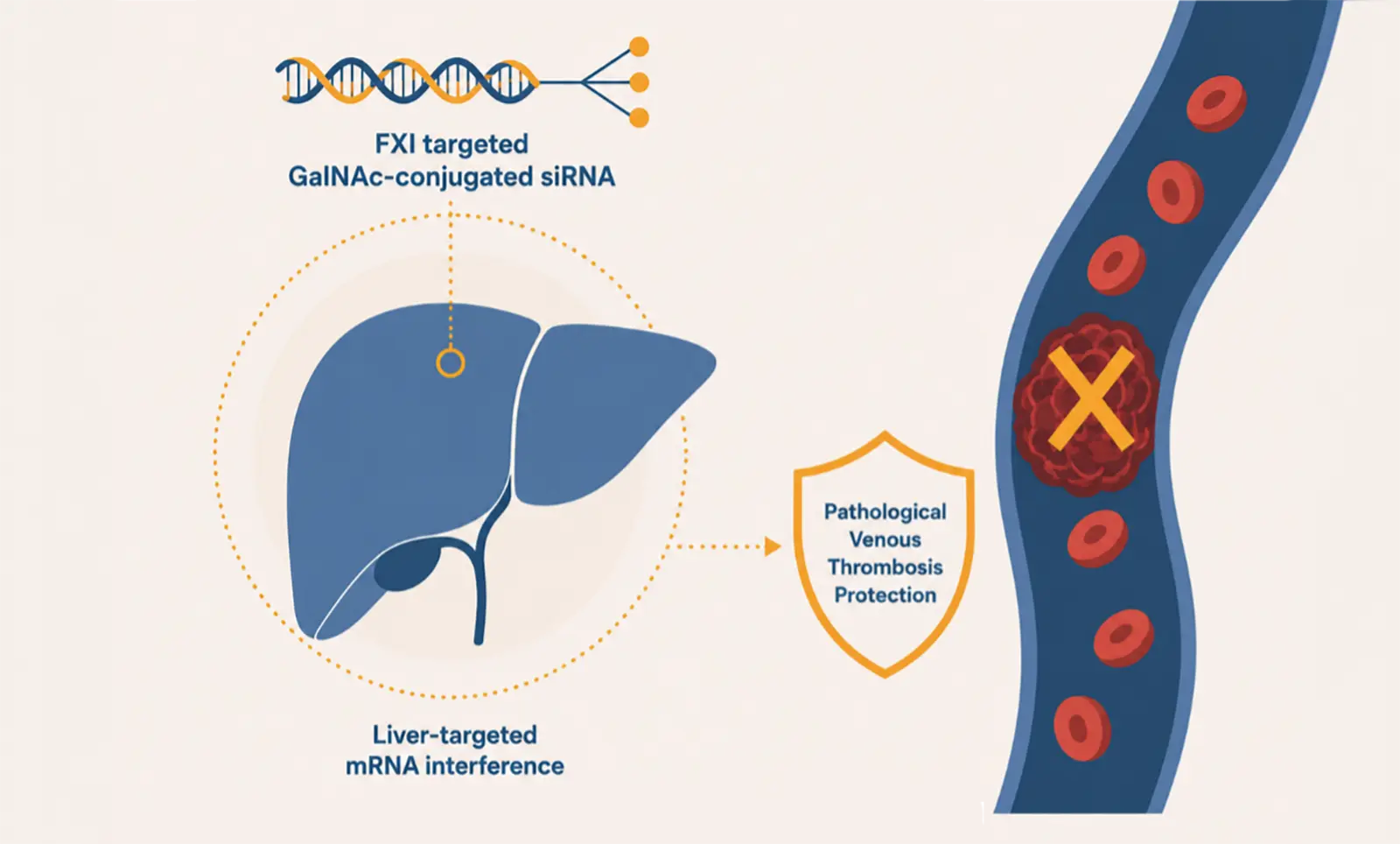
Suzhou Ribo Life Science (06938.HK) and its subsidiary Ribocure Pharmaceuticals AB (together “Ribo”) today announce the submission of a Phase 2b Clinical Trial Application (CTA) to the European Medicines Agency (EMA) for vortosiran (RBD4059), for prevention of venous thromboembolism (VTE).
Ribo is thereby expanding their anti-thrombotic clinical development of its lead siRNA asset, vortosiran (RBD4059), into the prevention of venous thromboembolism (VTE). This submission builds on Ribo’s experience across multiple thrombotic cardiovascular indications such as the Phase 2a study in coronary artery disease (CAD) and a recent Phase 2b Clinical Trial Application submission for stroke prevention in atrial fibrillation (SPAF). As the world’s most advanced FXI-targeting siRNA, vortosiran not only maintains a leading position in development progress but also demonstrates a broader therapeutic potential in future development.
Unlike current anticoagulants, which are associated with increased bleeding risk and require frequent dosing, vortosiran is designed to deliver durable antithrombotic protection with the potential for improved safety and infrequent administration. By selectively reducing Factor XI (FXI) levels, this approach aims to enable safer, long‑term VTE prevention, addressing key limitations of today’s standard of care.
”Our present CTA submission demonstrates our continued commitment to improve options for patients to prevent devastating thromboembolic disease and its complications. siRNA-mediated FXI inhibition may provide an optimized benefit-risk prevention profile for patients not receiving protection today” – says Dr. Anders Gabrielsen, Chief Medical Officer at Ribo.
VTE remains a major global health burden, affecting an estimated 10 million people worldwide each year and representing a leading cause of cardiovascular morbidity and mortality. A significant proportion of these patients are being undertreated or discontinuing therapy due to bleeding concerns associated with current anticoagulants. FXI inhibition via siRNA represents a novel therapeutic paradigm with the potential to improve the benefit-risk profile of antithrombotic therapy and expand access to safer long-term prevention for VTE patients.