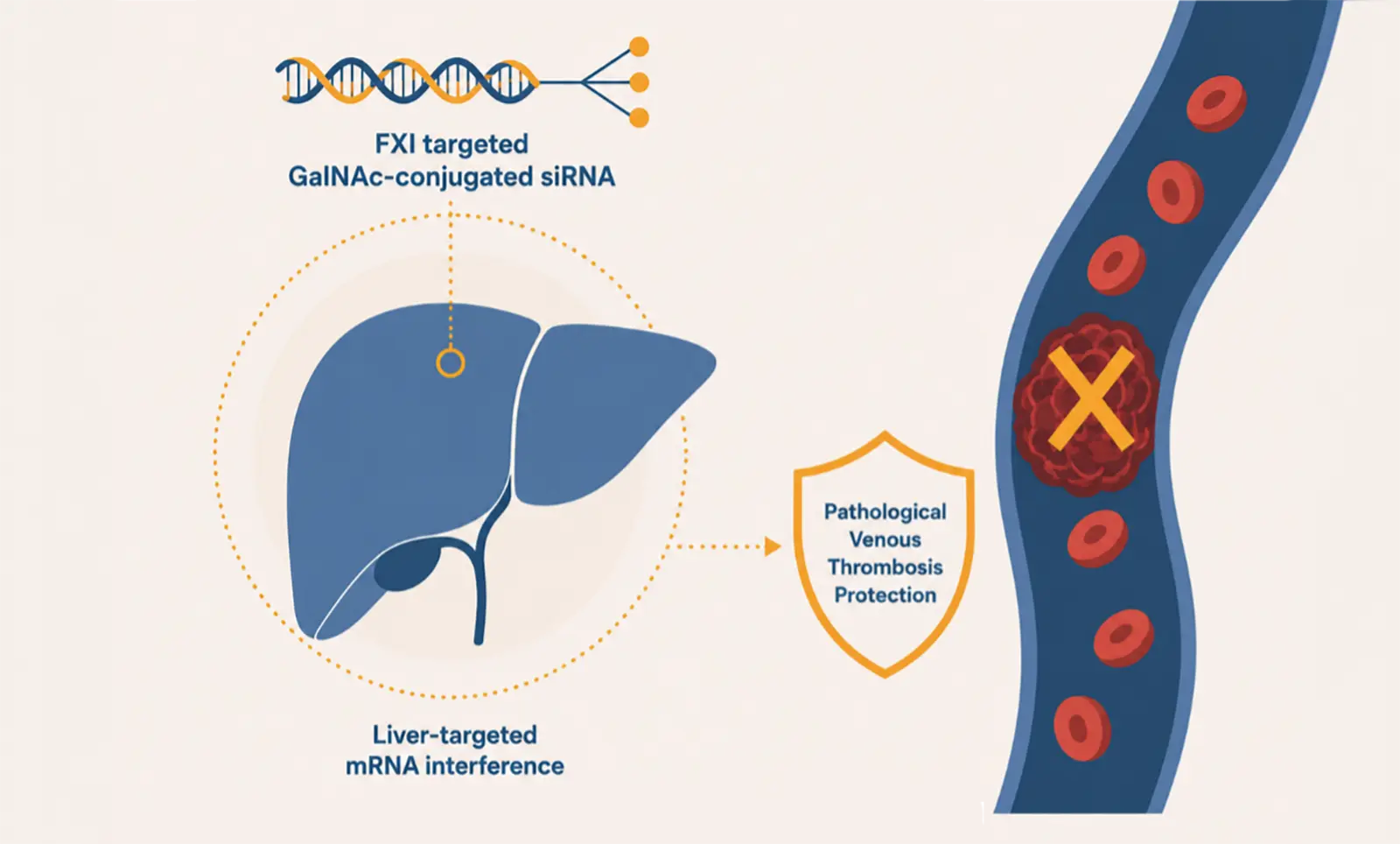
World’s first siRNA-based FXI silencing agent enters clinical development – an important step toward a new generation of anti-thrombotic therapies for our patients!
Suzhou Ribo announces the first siRNA-based FXI targeting anti-thrombotic agent (RBD4059) to receive HREC approval for first-in-human study in Australia.
On February 6th, 2023, Suzhou Ribo Life Science Co., Ltd. (hereinafter referred to as “Ribo”), a leading innovative oligonucleotide therapeutics company in China, announced that the first FXI (coagulation factor XI) targeting anti-thrombotic siRNA agent (RBD4059) had received HREC approval for its Phase 1 first-in-human clinical trial in Australia.
RBD4059, a GalNAc conjugated siRNA, is an FXI-targeted new molecular entity independently developed by Ribo using its proprietary siRNA delivery system – RIBO-GalSTAR®, and it is also the first FXI targeting molecule in the siRNA drug class that enters clinical stage. RBD4059 targets FXI mRNA and inhibits its expression in the liver and thus efficiently attenuates the activity of FXI in the blood, thereby blocks the intrinsic coagulation pathway and exerts its anticoagulant/anti-thrombotic effects. In non-clinical studies, RBD4059 demonstrated efficacious and long-lasting anti-thrombotic effects, as well as good safety profile, which supports its further clinical development.
The Phase I clinical trial, is a randomized, single-blind, and placebo-controlled study, to be conducted in healthy volunteers to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of single and multiple ascending doses of subcutaneously administered RBD4059. Results from this Phase I study will support upcoming global Phase II clinical trials in target patient populations.
Anticoagulation therapy is the mainstay of treatment and prevention of thromboembolic diseases. Patients with end-stage renal disease (ESRD), high risk coronary artery disease/peripheral arterial disease (CAD/PAD), atrial fibrillation (AF), and patients after orthopaedic surgery, etc, require routine anticoagulant therapy. Unfortunately, the existing anticoagulants, such as VKAs, heparins (unfractionated heparin and LMWH) and DOACs (FXa or FIIa inhibitors) are still associated with increased bleeding risk. Therefore, there is remaining major unmet medical need for developing drugs with high efficacy, good safety profile and long-lasting properties for the treatment and prevention of thromboembolic diseases.
Dr Li-Ming Gan, CMO and global R&D President of Ribo commented:
“There are increasing evidence supporting FXI inhibition to be an efficacious anti-thrombotic approach with low bleeding risk. FXI inhibition using RNA-interference approach is anticipated to add some crucial features, that will make RBD4059 a potential step change treatment for patients in need.”